THE HUMAN PLACENTA IS A CRUCIAL ORGAN TO ENSURE OPTIMAL FETAL GROWTH AND DEVELOPMENT
Earlier diagnosis of Placenta Dysfunction needed
The Centaflow Clinical System is designed to support placental dysfunction diagnostic by listening to the sound of abnormal blood flow. - We call it: The sound of turbulence.
Accurate
Non-invasive early detection of Placental Vascular Murmurs
Cost effective
Reduces total cost to healthcare providers while improving accuracy and reducing uncertainties.
Based on research
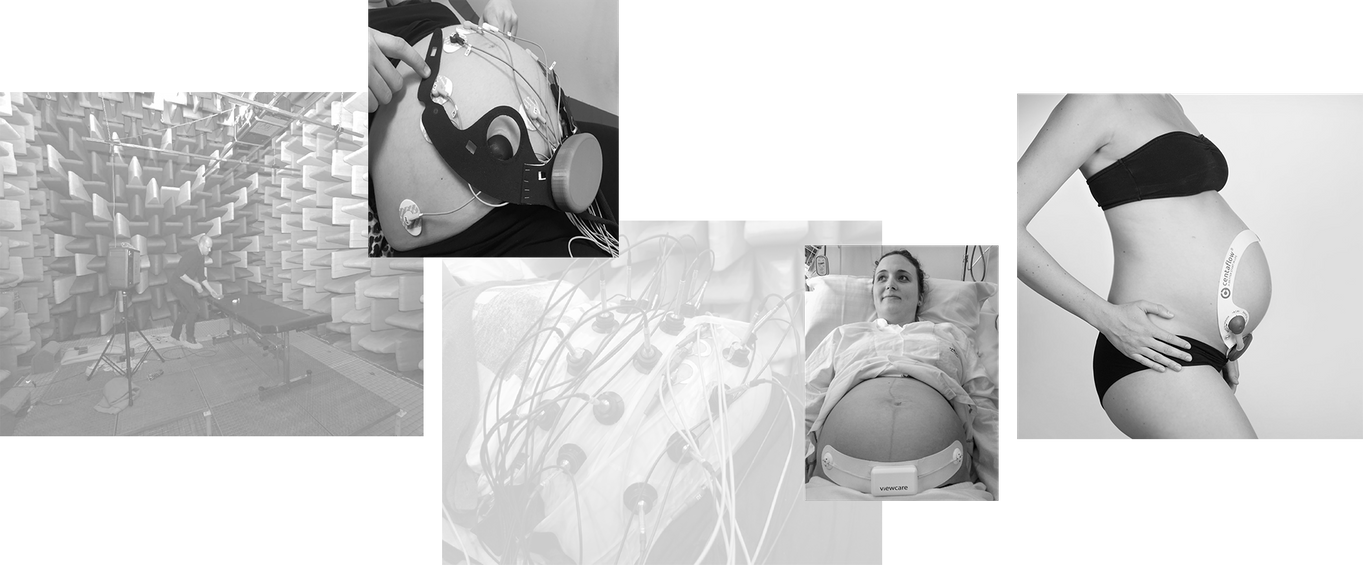
From silence
Our research efforts were initiated in an anechoic chamber which allowed us to isolate the sounds recorded from pregnant women. World-class danish acoustic expertise and experience has been the foundation for Centaflow since the early years.
→
To knowledge
We have made thousands of recordings of sounds emitted through the skin of the human abdomen.
Proven by scientific evidence
We are committed to scientific evidence and have conducted clinical trials with Centaflow since 2011.
Developed into a product
The Centaflow Clinical System is a non-invasive medical device for use by healthcare professionals as part of a panel of examinations used in standard care for pregnant women.
A perfected signal acquisition device
The device has been developed through several iterations that have all contributed significantly to the final design.
Safety and usability have been our main concerns every time we have improved the design.
Empowered by Artificial Intelligence
We work closely with universities and leverage the latest development in the field of Artificial Intelligence.
This allows us to derive precise and pertinent health-related information from the signals we gather from the human body.